Si vous êtes responsable de l’achat d’un nouveau système d’eau purifiée ou êtes responsable d’un système existant, il est important que vous compreniez ce que fait un système d’eau purifiée et comment les différentes composantes fonctionnent ensemble. Cet article vous donnera un aperçu des variations les plus courantes des systèmes de purification d’eau par osmose inversé (RO) pour les applications pharmaceutiques.
Flux de procédé général
La pharmacopée américaine (et d’autres) spécifie que l’eau purifiée doit être produite à partir d’une source d’eau potable. Pour simplifier l’article, nous supposons ici que le système d’eau purifiée est alimenté à partir d’une source d’eau potable municipale.
Diagrammes du système de production d'eau purifiée pharmaceutique
Les schémas ci-dessous illustrent les principaux composants typiques des systèmes de production et de distribution d'eau purifiée pharmaceutique. Poursuivez votre lecture pour obtenir une explication détaillée du fonctionnement de chaque unité.
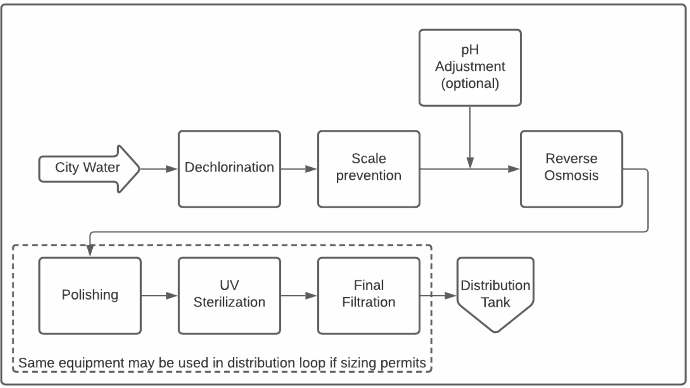
Boucle de distribution d'eau purifiée typique
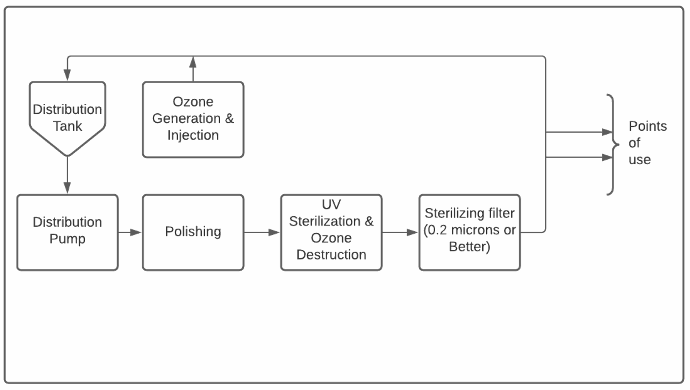
Osmose inversé – Le cœur du système
Au cœur de presque tous les systèmes modernes de purification de l'eau se trouve un appareil d'osmose inverse (OI). L'osmose inverse est un procédé qui permet d'éliminer les impuretés dissoutes dans l'eau en forçant l'eau à traverser une membrane semi-perméable. L'eau est débarrassée de la plupart de ses contaminants au fur et à mesure qu'elle traverse la membrane. Cette eau purifiée est communément appelée perméat d'osmose inverse.
Cependant, toute l'eau ne traverse pas la membrane. Une partie de l'eau reste du côté "sale" de la membrane et contribue à évacuer les contaminants de la surface de la membrane. Pour éviter le colmatage de la membrane, deux conditions doivent être remplies :
- Les contaminants doivent être maintenus en solution ; et
- Les contaminants doivent être emportés par l'eau, ce qui signifie que le "flux transversal" doit être maintenu pour favoriser les turbulences et les vitesses appropriées à la surface de la membrane.
Un système d'osmose inverse envoie généralement environ 25 % de son eau d'alimentation au drain. Cela signifie que 4 gallons d'eau d'alimentation sont nécessaires pour produire 3 gallons de perméat d'osmose inverse. Ce rapport de 3:4 signifie que le système fonctionne avec un taux de récupération de 75 %. Les taux de récupération du système sont limités par les choix de conception de l'OI et les conditions de fonctionnement.
Pré-traitement : faire battre le cœur
Les performances des machines RO peuvent se dégrader dans les conditions suivantes :
- Encrassement inorganique de la surface de la membrane ;
- Encrassement organique de la surface de la membrane ;
- Encrassement biologique de la surface de la membrane ;
- Oxydation de la membrane ; ou
- Obstruction de l’écoulement par des sédiments ou d’autres solides en suspension
- Dégradation normale de la matrice de la membrane due à l'âge et à l'exposition aux produits chimiques lors des nettoyages.
Dégradation normale de la matrice de la membrane due à l'âge et à l'exposition aux produits chimiques lors des nettoyages.
L’encrassement organique est causé par la présence de composés organiques dans l’eau d’alimentation. Il est parfois évité par une étape de pré-traitement distincte et d’autres fois traitées en effectuant des nettoyages périodiques à pH élevé de l’osmose inversée.
L’encrassement biologique se produit lorsque les bactéries recouvrent une partie importante de la surface de la membrane avec un biofilm, empêchant le flux à travers la membrane. L’encrassement biologique peut être ralenti par une désinfection en amont et en maintenant un peu d’oxygène dissous dans l’eau d’alimentation. Les systèmes pharmaceutiques RO sont généralement conçus pour être désinfectés chimiquement ou avec de l’eau chaude sur une base régulière afin de limiter la croissance bactérienne.
L’oxydation des membranes est rapide et irréversible. Elle est causée par la présence d’oxydants dans l’eau d’alimentation, tels que le chlore, les chloramines ou l’ozone. La plupart des systèmes d’eau purifiée comprennent une étape de déchloration, car la plupart des approvisionnements en eaux municipaux fournissent de l’eau avec un résidu de chlore et/ou de chloramine.
Les sédiments et autres solides en suspension "bouchent" physiquement les membranes d'osmose inverse dans un système. Cela entraîne un débit réduit et irrégulier à travers les éléments de la membrane, augmentant le taux d'encrassement, diminuant la production d'eau et finalement la qualité de l'eau purifiée.
Polissage
Dans la plupart des cas, le perméat d’une RO n’est pas assez purifié pour répondre aux normes des pharmacopées. L’étape de polissage élimine en outre les ions et les matières organiques dissous, aidant à respecter de manière fiable les limites de conductivité et de carbone organique total (COT). Ceci est parfois réalisé avec une deuxième étape de purification RO, souvent appelée une « deuxième passe ».
Électrodésioniseur (EDI) stérilisable à la chaleur installé sur un système de production d’eau purifiée

Distribution
L’eau purifiée ne reste pas pure très longtemps. Après tout, l’eau est le solvant universel ainsi que la base de la vie telle que nous la connaissons! Les systèmes de distribution d’eau purifiée sont le plus souvent conçus comme des boucles où l’eau purifiée est stockée dans un réservoir sanitaire et est pompée autour d’une boucle avec un ou plusieurs points d’utilisation sur demande.
Bien que les conceptions des boucles de distribution varient, il est largement admis qu’elles devraient maintenir une vitesse minimale de l’eau et ne contenir aucun volume mort – des zones où l’eau peut stagner en dehors du débit principal.
L’eau circulant à travers la boucle de distribution retourne parfois à travers l’étape de polissage afin de la maintenir purifiée car l’air dans le réservoir de stockage provoque une augmentation de la conductivité lorsque le CO2 se dissout dans l’eau.
Stérilisation
La dernière étape de la production d’eau purifiée est la stérilisation. Cette étape est généralement intégrée dans la boucle de distribution. Bien que de nombreuses approches existent, presque tous les systèmes comprennent une étape de filtration avec des filtres à cartouche de 0,2 ou 0,1 micron (µm) fournissant une barrière physique finale contre les bactéries. De plus, de nombreux systèmes comprennent un système de désinfection à l’ozone pour maintenir de faibles niveaux d’ozone dans le réservoir de distribution et, parfois, pour permettre une stérilisation périodique à l’ozone de la boucle de distribution elle-même.
Enfin, les unités de stérilisation aux ultraviolets (UV) sont souvent utilisées pour la stérilisation et, parfois, pour la réduction du COT. Il est généralement recommandé que ces unités soient installées en amont du filtre final afin que les bactéries mortes ou désactivées soient capturées par le filtre. Les UV sont également utilisés pour décomposer l’ozone puisque les eaux officinales ne doivent pas contenir d’ozone au point d’utilisation.
Finally, ultraviolet (UV) sterilization units are often used for sterilization and, sometimes, for TOC reduction. It is generally recommended that these units be installed upstream of the final filter so that any dead or deactivated bacteria will be caught by the filter. UV is also used to break down ozone since compendial waters must not contain ozone at the point of use.
Conclusion
Vous avez des questions sur le fonctionnement des systèmes de purification d’eau pharmaceutiques? Contactez-nous ou laissez un commentaire ci-dessous. Nous aimerions recevoir de vos nouvelles!