Quelle stratégie de prévention de l'entartrage convient le mieux à votre système d'eau pharmaceutique ?
La technologie de l'osmose inverse (OI) est désormais omniprésente dans la purification de l'eau. Alors que les systèmes plus anciens reposaient parfois sur la distillation comme principale stratégie de purification, l'OI est devenue le cheval de bataille standard, même pour la production d'eau pour injection (WFI) dans le cadre de la plupart des pharmacopées. Besoin d'un rappel sur le fonctionnement des systèmes de purification de l'eau ? Consultez notre article ici.
L'osmose inverse est une technologie fantastique lorsqu'elle est appliquée correctement. Elle est cependant assez sensible et il est important de s'assurer que l'eau que vos membranes d'OI purifient ne va pas les endommager ou les encrasser (les boucher). Les membranes d'OI sont assez facilement colmatées par le tartre, qui est causé par la dureté dépassant ses limites de saturation plutôt basses du côté "sale" de la membrane d'OI. C'est pourquoi le prétraitement OI comprend généralement une étape visant à éliminer la dureté ou à augmenter sa limite de saturation.
Après avoir lu cet article, vous devriez avoir une idée relativement claire de la technologie la mieux adaptée à votre situation. Nous allons comparer trois technologies : les adoucisseurs, le dosage de produits chimiques antitartre et l'élimination électrolytique du tartre (ESR).
Facteurs pris en compte
Pour chaque technologie, nous avons évalué le coût d'investissement, les frais d'exploitation directs, la fiabilité, les effets négatifs en aval, les exigences en matière de maintenance et d'exploitation et l'empreinte écologique. En fonction de vos priorités, chacun de ces facteurs sera plus ou moins important pour vous. C'est pourquoi nous vous recommandons vivement de procéder à une analyse approfondie de vos besoins avant de choisir une technologie de traitement.
Nous présentons les résultats de notre comparaison dans le tableau récapitulatif ci-dessous et expliquons notre raisonnement pour tous les critères, à l'exception de l'empreinte, dans les sections suivantes.
Résumé
|
|
Adoucisseur |
Antiscalant |
ESR |
|
Coût du capital |
$$ |
$ |
$$$$ |
|
Coût d'exploitation annuel |
$$$$ |
$$$ |
$ |
|
Fiabilité |
|
|
|
|
Impacts négatifs en aval |
XX |
XXXX |
X |
|
Exigences en matière d'O&M |
+++ |
++ |
+ |
|
Empreinte à 5 US GPM |
6’x2’ |
3’x3’ |
5’x5’2 |
|
Empreinte à 80US GPM |
8’x12’ |
3’x3’ |
6’x11.5’ |
Comparaison de haut niveau entre l'adoucissement, l'anticalcaire et l'ESR
1 Nous avons supposé que l'utilisation de l'agent antitartre entraînera un remplacement plus fréquent des membranes. Pour les estimations OpEx excluant les coûts de remplacement des membranes, se référer à la section détaillée des dépenses d'exploitation.
2 L'encombrement de l'ESR est notre meilleure estimation prudente sur la base des fiches techniques disponibles, qui concernent des systèmes complets d'eau purifiée et de WFI. L'encombrement réel peut être inférieur.
Fiabilité
Les trois options présentées ici sont relativement fiables. Lors de l'évaluation de la fiabilité, nous avons pris en compte à la fois la fiabilité de l'équipement lui-même et sa sensibilité aux variations de la qualité de l'eau d'alimentation.
Fiabilité des adoucisseurs d'eau
Les adoucisseurs d'eau sont une technologie très ancienne, qui a fait ses preuves. S'ils sont correctement entretenus, ils fonctionnent généralement très bien et tombent rarement en panne. En outre, ils peuvent être réglés pour traiter de manière fiable le "pire cas" de qualité de l'eau d'alimentation sans provoquer d'effets négatifs en aval.
Ils sont cependant sujets à des défaillances mécaniques, en particulier lorsque les tâches d'entretien ne sont pas effectuées avec suffisamment de fréquence ou de diligence. Par exemple, le système de saumure doit être nettoyé régulièrement pour éviter les obstructions et autres dysfonctionnements qui feraient que l'adoucisseur transmettrait de l'eau dure au système d'osmose inverse. Les adoucisseurs d'eau sont souvent équipés de vannes complexes à orifices multiples ou de nids de vannes comportant un grand nombre de joints et de pièces mobiles susceptibles de tomber en panne. Malgré ces points de défaillance possibles, une maintenance diligente, très courante dans l'industrie pharmaceutique, permet généralement aux adoucisseurs d'eau de fonctionner de manière très fiable.
La fiabilité des adoucisseurs est renforcée par le fait qu'ils éliminent efficacement d'autres cations tels que le fer, qui peut être un problème pour les osmoseurs inversés protégés par un antitartre et un ESR.
Fiabilité de l'antitartre
La chimie de l'antitartre fonctionne très bien dans les limites des paramètres de qualité de l'eau spécifiés. Cela dit, les systèmes de dosage de l'antitartre dépendent d'une qualité relativement stable de l'eau d'alimentation, en particulier du pH et de la dureté. Contrairement aux adoucisseurs, un dosage incorrect dû à une qualité variable de l'eau d'alimentation peut entraîner un encrassement prématuré des membranes d'osmose inverse.
Comme pour tout système de dosage de produits chimiques, un entretien régulier de la (des) pompe(s) doseuse(s), des tuyaux et des vannes est nécessaire pour éviter les colmatages et autres dysfonctionnements.
Fiabilité de l'ESR
La réduction électrolytique du tartre est de loin l'option qui comporte le moins de pièces mobiles. Pour cette raison, on peut raisonnablement s'attendre à ce qu'un système ESR correctement dimensionné pour la qualité et le débit de l'eau d'alimentation fonctionne bien tant que l'entretien recommandé est effectué.
Malheureusement, notre équipe ne dispose pas d'expérience directe ou de données clients sur la fiabilité de la technologie ESR. Nous nous sommes basés sur des témoignages filmés de clients, sur les réactions de clients utilisant l'ESR dans des applications de tours de refroidissement et sur une analyse technique de la technologie pour déterminer la fiabilité attendue.
Cette expérience nous a permis de comprendre que l'ESR est très fiable lorsqu'il est utilisé dans le cadre de paramètres de conception relativement étroits. Si la qualité de l'eau, y compris le pH et la dureté, a tendance à varier considérablement dans l'eau d'alimentation, nous pensons qu'un encrassement prématuré de la membrane d'OI peut en résulter. Ceci étant dit, l'ESR dimensionné pour le pire scénario serait probablement la solution la plus fiable pour la réduction du tartre avant OI disponible aujourd'hui.
Impacts négatifs en aval
Si les technologies de prétraitement existent évidemment pour protéger les équipements de purification en aval, ces trois technologies modifient considérablement la chimie de l'eau et il est donc important d'être conscient des effets potentiellement négatifs qu'elles ont sur le système d'OI et sur la qualité de l'eau qu'il produira.
Impacts négatifs en aval des adoucisseurs d'eau
Les adoucisseurs remplacent la plupart des cations divalents et trivalents de l'eau d'alimentation par des ions sodium monovalents. Bien que ce phénomène soit relativement bénin, il est important car les membranes d'osmose inverse ont beaucoup plus de facilité à rejeter les ions divalents et trivalents que le sodium. En bref, cela signifie que l'on peut s'attendre à ce que l'adoucissement augmente la conductivité du perméat d'osmose inverse proportionnellement à la charge en cations non sodiques de l'eau d'alimentation.
Dans la plupart des cas, la quantité de sodium ajoutée à l'eau d'alimentation n'est pas suffisamment importante pour causer un réel problème de procédé. Cependant, lors de la purification d'une eau fortement chargée (conductivité élevée), il peut être intéressant d'explorer d'autres options si la conductivité de l'osmose inverse au premier passage est un facteur de conception limitatif.
Impacts négatifs en aval de l'antitartre
Bien que le dosage de l'agent antitartre soit l'une des approches les plus populaires de la prévention de l'entartrage dans les systèmes d'OI de grande taille (>30 US GPM), c'est aussi la méthode que nous avons vue le plus souvent conduire à une courte durée de vie de la membrane. Un examen de la littérature scientifique révèle que le dosage de l'agent antitartre doit être très précis pour être très bénéfique. Un sous-dosage de l'antitartre peut évidemment entraîner l'encrassement des membranes par le tartre (dépôts de dureté ou de métal sur la membrane), tandis qu'un surdosage peut entraîner le dépôt de l'antitartre lui-même sur la membrane. Il a également été observé que l'utilisation d'antitartres à base de polymères augmente de manière significative l'encrassement biologique.
En bref, si les produits antitartre sont la solution la moins coûteuse, c'est celle qui cause généralement le plus de maux de tête et de coûts en aval.
Impacts négatifs de l'ESR en aval
Outre l'impact négatif évident d'un système ESR mal dimensionné - l'entartrage de la membrane dû à une fuite importante de dureté - nous n'avons pas pu identifier d'autres effets négatifs suspects en aval. Il convient de noter que le système ESR élimine activement les contaminants du flux d'eau, ce qui facilite le travail de l'OI au-delà de la simple prévention de l'entartrage des membranes.
Un petit détail que nous avons noté est que le fabricant d'ESR BioPureMax indique qu'une certaine quantité de chlore libre est générée en très petites quantités par les systèmes ESR(source). Nous ne pensons pas qu'il s'agisse d'un problème important, car nous nous attendons à ce que l'étape de l'ESR soit suivie d'une étape de déchloration dans tous les systèmes où elle est installée..
En outre, les fabricants de technologies ESR affirment que la technologie élimine la dureté pour empêcher efficacement l'entartrage des membranes d'OI. Nous serions prudents quant à l'utilisation de l'ESR lors de la conception de systèmes d'OI à récupération élevée qui concentrent le flux d'alimentation au-delà de la concentration 4x observée dans les systèmes fonctionnant à une récupération relativement standard de 75%.
Exigences en matière d'exploitation et de maintenance
Dans cette section, nous examinons dans quelle mesure l'intervention de l'opérateur est nécessaire pour exploiter et entretenir correctement chacune des trois options de prévention de l'entartrage. Nous couvrons ces exigences séparément des frais d'exploitation liés à chaque option, car certains clients considèrent que certaines exigences sont particulièrement onéreuses, indépendamment de leurs implications en termes de coûts.
Exigences en matière d'exploitation et de maintenance pour les adoucisseurs
L'opération la plus évidente pour les systèmes d'adoucissement est le chargement du sel dans le réservoir de saumure de l'adoucisseur. Dans la plupart des systèmes de purification de l'eau pharmaceutique, il s'agit d'une opération manuelle plutôt que d'un processus assisté par une machine ou automatisé comme dans les usines dotées de grands systèmes centraux de saumure.
Outre la nécessité de remplir le réservoir de saumure, généralement sur une base hebdomadaire, les adoucisseurs doivent être surveillés pour détecter les fuites de dureté, de préférence à l'aide d'analyseurs de dureté en ligne. D'autres tâches d'entretien telles que le nettoyage du réservoir de saumure et de la vanne de saumure, la vérification du fonctionnement des cycles de régénération et le test de la capacité de la résine ne doivent être effectuées qu'une fois par an ou tous les six mois.
Dans l'ensemble, les adoucisseurs nécessitent le plus d'interventions de la part de l'opérateur et certainement les travaux les plus lourds (au sens propre) des trois options de prévention de l'entartrage. Cela dit, ils restent très faciles à gérer pour la plupart des usines pharmaceutiques.
Exigences en matière de fonctionnement et d'entretien des systèmes d'anti-calcaire
Les systèmes de dosage d'antitartre nécessitent un contrôle régulier de la dureté à l'entrée et des ajustements au niveau de la pompe doseuse lorsque les niveaux de dureté fluctuent. Sinon, ces systèmes nécessitent relativement peu d'entretien, une inspection visuelle rapide étant nécessaire pour s'assurer qu'il n'y a pas de bouchons ou de fuites et que l'antitartre s'écoule comme prévu.
Certains opérateurs préfèrent utiliser le moins possible de produits chimiques dangereux. Dans ce cas, l'antitartre peut être une option à éviter, malgré l'intervention relativement limitée de l'opérateur pour remplacer périodiquement les seaux ou les tonneaux. Les produits chimiques sont également relativement faciles à manipuler.
Exigences en matière d'exploitation et de gestion pour l'ESR
Les systèmes ESR fonctionnant selon les paramètres de conception spécifiés nécessitent très peu d'entretien. En général, ils sont autonettoyants, inversent le courant et évacuent automatiquement le précipité accumulé quotidiennement. Ces systèmes peuvent nécessiter un simple nettoyage avec un nettoyeur haute pression environ une fois par an pour éviter toute perte de performance. De plus, il est possible de contrôler leur efficacité et la nécessité d'un nettoyage en surveillant leur consommation d'énergie. Ils consomment plus d'énergie car la résistance entre l'anode et la cathode diminue avec l'accumulation de tartre.
Comparaison des coûts
Tous les calculs OpEx sont basés sur un fonctionnement 24/7/365 et 10 grains par gallon de dureté.
CAPEX
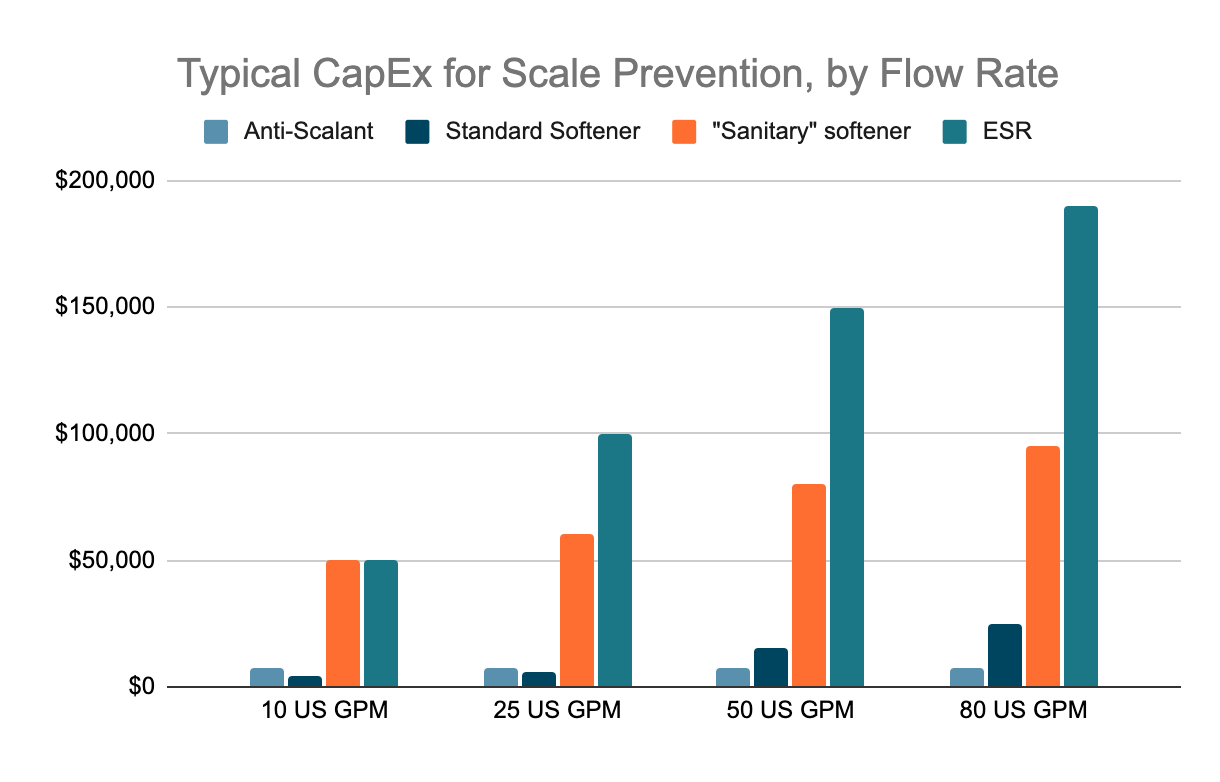
OPEX
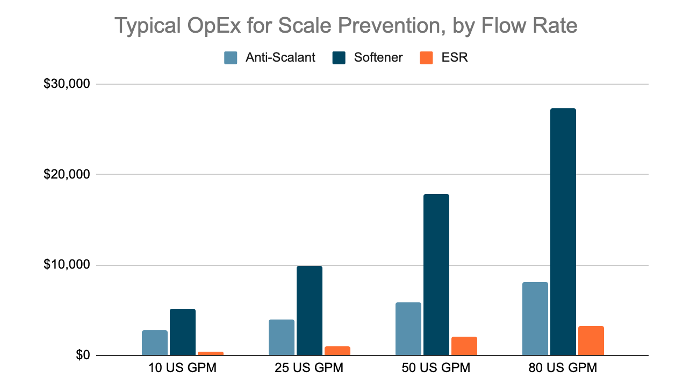
L'OpEx de l'adoucisseur est basé sur deux visites d'entretien à 1 000 $ chacune et un coût de 6 $ par sac de sel de 18,1 kg (39,8 lb), un dosage de 10 lb de sel par pied cube de résine et une capacité de 25 000 grains par pied cube de résine.
Antiscalant OpEx basé sur un dosage de 4,5 mg/L d'Hypersperse MDC700 de SUEZ au prix de 8,57 $/kg et 2 000 $ de coûts annuels de maintenance de la pompe en pièces et main d'œuvre.
ESR OpEx basé sur des coûts d'électricité de 0,09 $ / kWh.
Pour plus d'informations sur le coût des systèmes pharmaceutiques de purification de l'eau, lisez notre article ici.
Conclusion
Choisir la meilleure solution de prévention du tartre pour votre système d'eau purifiée n'est pas une décision simple. Il n'existe tout simplement pas de "meilleure" technologie, bien qu'il y ait presque toujours un "meilleur choix" pour une application donnée lorsqu'une évaluation approfondie des besoins, incluant les facteurs techniques, économiques, environnementaux et humains, a été réalisée.
Quelles sont vos meilleures pratiques en matière de prévention du tartre pour les systèmes d'eau pharmaceutique ? Participez à la conversation en laissant un commentaire ci-dessous. Nous serions ravis d'entendre ce que vous avez à dire !